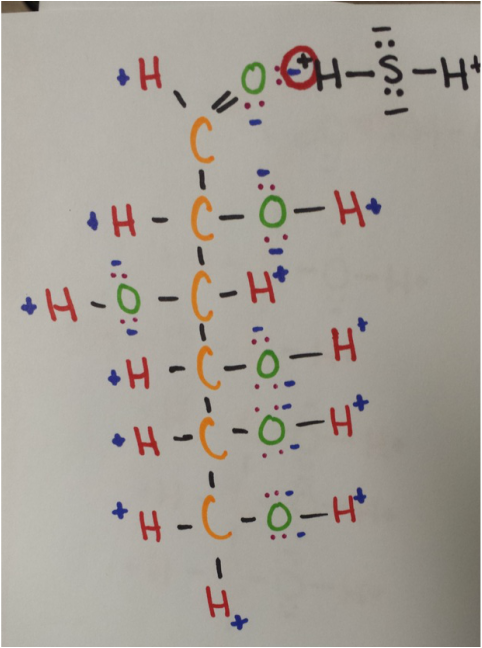
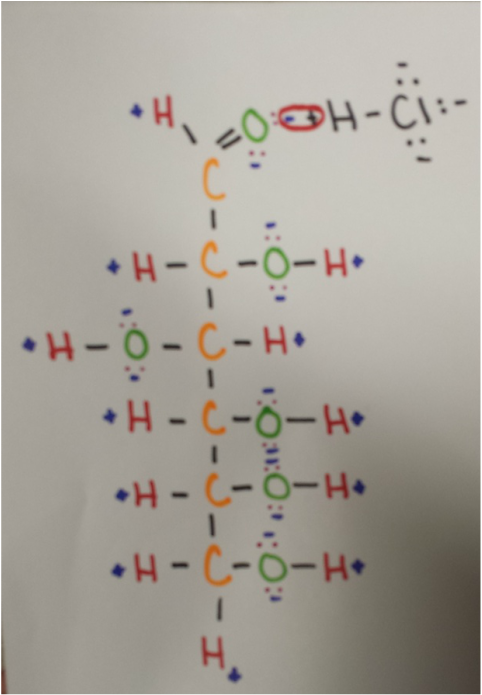
Intermolecular Forces: C6H12O6 and H2S
Dispersion forces:
In this case, dispersion forces are acting on these two molecules. Dispersion forces always act on two molecules that are adjacent to one another.
Dipole-Dipole:
Linear glucose and hydrogen sulfide have a dipole-dipole attraction because both are polar molecules. The positive hydrogen atom in the H2S is attracted to the negative oxygen atom of C6H12O6.
Hydrogen Bonding:
Hydrogen bonding is occurring because the negative oxygen of one molecule (linear glucose) is attracted to the hydrogen atom of another molecule (hydrogen sulfide).
In this case, dispersion forces are acting on these two molecules. Dispersion forces always act on two molecules that are adjacent to one another.
Dipole-Dipole:
Linear glucose and hydrogen sulfide have a dipole-dipole attraction because both are polar molecules. The positive hydrogen atom in the H2S is attracted to the negative oxygen atom of C6H12O6.
Hydrogen Bonding:
Hydrogen bonding is occurring because the negative oxygen of one molecule (linear glucose) is attracted to the hydrogen atom of another molecule (hydrogen sulfide).
Intermolecular Forces: C6H12O6 and HCl
Dispersion forces:
Dispersion forces are acting on the linear glucose and hydrogen chloride because they are two adjacent molecules, and dispersion forces always act upon adjacent molecules.
Dipole-Dipole:
Dipole-dipole forces are acting upon these two molecules because both are polar. The positive hydrogen atom of HCl is attracted to the negative oxygen atom of linear glucose.
Hydrogen Bonding:
Between these two molecules, hydrogen bonding is occurring, because the hydrogen of hydrogen chloride is attracted to the oxygen of C6H12O6.
Dispersion forces are acting on the linear glucose and hydrogen chloride because they are two adjacent molecules, and dispersion forces always act upon adjacent molecules.
Dipole-Dipole:
Dipole-dipole forces are acting upon these two molecules because both are polar. The positive hydrogen atom of HCl is attracted to the negative oxygen atom of linear glucose.
Hydrogen Bonding:
Between these two molecules, hydrogen bonding is occurring, because the hydrogen of hydrogen chloride is attracted to the oxygen of C6H12O6.